Molecular Orbitals Q2
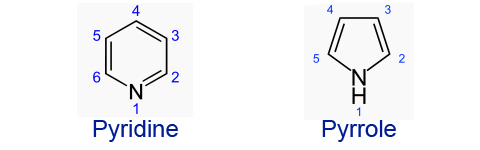
Heterocyclic amines are a very important type of amino group. One use of them includes being the base of DNA. The most important property however is their basicity due to nitrogen.
Pyridine is a much stronger base than pyrrole because:
[hide-this-part morelink=”Reveal Answer”]
D.
In pyrrole, the sigma bonds around nitrogen are formed by sp2 hybrid orbitals, not sp3. A nitrogen electron participates in each of the sigma bonds. The remaining nitrogen electrons are in a nitrogen 2p orbital which overlaps with the carbon 2p orbitals in the ring to form pi bonds and the cyclic compound, delocalizing the lone pair. In pyridine, the nitrogen atom donates one electron to each of the two sigma bonds by use of sp2 orbitals. The third sp2 orbital contains two electrons, which are for acids. One electron of nitrogen is present in a p orbital, which interacts with the carbon p orbitals to form the pi bond.
[/hide-this-part]