[php snippet=1]
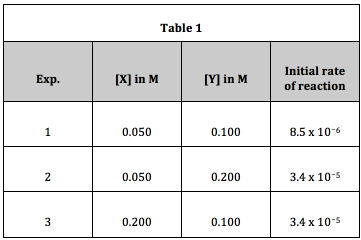
The data in Table 1 were collected for Reaction I:
2X + Y –> Z
Reaction I
What is the rate law for the reaction?

Loading ...
Correct Answer: C. Rate = k[X][Y]2
This is a common MCAT-type question. By looking at Table 1, we can see that when the concentration of X is quadrupled (factor of 41) while [Y] is unchanged (Exp. 1 and 3), the rate is increased by a factor of 4 = 41. Thus the order of the reaction with respect to X is 1 (= “first order with respect to X”).
When the concentration of Y is doubled (factor of 21) while [X] remains the same (Exp. 1 and 2), the rate of reaction is quadrupled (factor of 4 = 22). Thus, the order of reaction with respect to Y is 2 (= “second order with respect to Y”).
The rate equation is Rate = [X][Y]2. Since the overall rate of reaction is the sum of exponents, we can say that the reaction is (1 + 2) a third order reaction overall.
{Notice that the stoichiometric coefficients are not relevant; the order of reaction being based on data.}
Additional learning point: In Organic Chemistry, an SN2 reaction is typical of a second order reaction where the rate must be equally proportional to the concentration of both reactants; for example, first order with respect to the alkyl halide and first order with respect to the nucleophile, so overall second order.
Today’s question has been brought to you by Gold Standard MCAT.
Get more passage-based questions at Gold Standard MCAT.
Subscribe below to receive the MCAT Question of the Day delivered straight to your inbox every morning.

