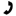Van der Waals Real Gas Equation

Which of the following statements about van der Waals equation are true?
I. At low densities, a real gas behaves like an ideal gas.
II. The “b” term in the equation describes intermolecular forces.
III. The product PV equals a constant for real and ideal gases alike.
Subscribe below to receive the MCAT Question of the Day delivered straight to your inbox every morning.
Photo attributed to edenpictures.