Boyle’s Law
Boyles law is a special case of the ideal gas law in which P1V1 = P2V2, stating that the volume of a gas is inversely proportional to pressure at a constant temperature. Which of these gases correctly demonstrates Boyles Law?
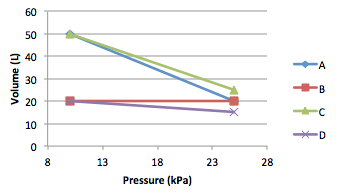
Boyles law is a special case of the ideal gas law in which P1V1 = P2V2, stating that the volume of a gas is inversely proportional to pressure at a constant temperature. Which of these gases correctly demonstrates Boyles Law?
