Electrons In An Atom

What is the maximum number of electrons in an atom that can have n = 3, l = 2 quantum numbers?
Subscribe below to get the MCAT question of the day sent straight to your inbox!
Photo attributed to Germanno.

What is the maximum number of electrons in an atom that can have n = 3, l = 2 quantum numbers?
Photo attributed to Germanno.
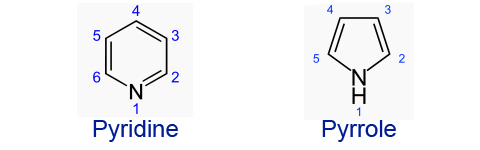
Heterocyclic amines are a very important type of amino group. One use of them includes being the base of DNA. The most important property however is their basicity due to nitrogen.
Pyrrole undergoes electrophilic substitution at position 2 because:
[hide-this-part morelink=”Reveal Answer”]
A.
Attack at position 2 gives three resonance structures greatly adding to stability of the molecule (positive charge on nitrogen, then position 3, and then position 5). Position 3 only has two resonance structures.
[/hide-this-part]
[wp_email_capture_form]

Heterocyclic amines are a very important type of amino group. One use of them includes being the base of DNA. The most important property however is their basicity due to nitrogen.
Pyridine is a much stronger base than pyrrole because:
[hide-this-part morelink=”Reveal Answer”]
D.
In pyrrole, the sigma bonds around nitrogen are formed by sp2 hybrid orbitals, not sp3. A nitrogen electron participates in each of the sigma bonds. The remaining nitrogen electrons are in a nitrogen 2p orbital which overlaps with the carbon 2p orbitals in the ring to form pi bonds and the cyclic compound, delocalizing the lone pair. In pyridine, the nitrogen atom donates one electron to each of the two sigma bonds by use of sp2 orbitals. The third sp2 orbital contains two electrons, which are for acids. One electron of nitrogen is present in a p orbital, which interacts with the carbon p orbitals to form the pi bond.
[/hide-this-part]
[jwplayer config=”Youtube” file=”http://www.youtube.com/watch?v=ginsUQJmHP4″]
This question requires the use of a periodic table.
[hide-this-part morelink=”Reveal Answer”]
D.
A diamagnetic ion will have no unpaired subshells in its atomic orbitals. A paramagnetic ion will have one unpaired electron in its atomic orbitals, leading it to be slightly more magnetic due to the unbalance. For example, Zn2+ will lose 2 electrons from the 4s2 while leaving the 3d10 orbital full, leaving no unpaired electrons. Copper will lose electrons from the 4s2 orbital to fill the 3d10 orbital, a more stable configuration and thus a diamagnetic compound.
[/hide-this-part]

A reaction is found to have an equilibrium constant of 600 at a temperature of 300K and a pressure of 1.0 atm. It can be concluded that at these conditions:
I. ΔH < 0 II. ΔS < 0 III. ΔG < 0
[hide-this-part morelink=”Reveal Answer”]
C.
Since K > 0, the reaction is spontaneous, and ΔG < 0; ΔG = -RTln(k) Although ΔH and ΔS combine to determine ΔG, we lack information to determine the sign of either of them. [/hide-this-part]

Acetic acid reacts more rapidly with methanol than with ethanol to form an ester. Most likely this is due to:
[hide-this-part morelink=”Reveal Answer”]
C.
Bulky groups on either the acid or alcohol slow the reaction due to steric hindrance.
[/hide-this-part]

For those atoms in the first column of the periodic table which form stable positive ions, the ion usually has a considerably smaller radius than the neutral atom. What explains this effect?
[hide-this-part morelink=”Reveal Answer”]
D.
The electron in the neutral atom occupies an s orbital of higher n than the valence electrons of the positive ion. The former electron will stay, on average, farther from the nucleus than the inner electrons.
[/hide-this-part]
Which of the following has (have) buffer properties?
I. 50 mL of .1 M HCl
II. 100 mL of .1 M acetic acid
III. 50mL of .1 M NaOH
[hide-this-part morelink=”Reveal Answer”]
D.
By mixing a weak acid with NaOH, we convert some of the acetic acid to acetate. The resulting mixture of the acid and its conjugate base is a buffer, and [H+] will be within an offer of magnitude of Ka.
[/hide-this-part]
[wp_email_capture_form]