Air Dissolved in Water
A sealed vessel holds air and liquid water. Which of the following conditions would greatly increase the amount of air dissolved into the water?
A sealed vessel holds air and liquid water. Which of the following conditions would greatly increase the amount of air dissolved into the water?
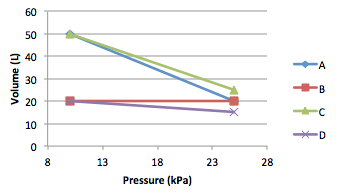
Boyles law is a special case of the ideal gas law in which P1V1 = P2V2, stating that the volume of a gas is inversely proportional to pressure at a constant temperature. Which of these gases correctly demonstrates Boyles Law?
Which of the following is the most likely to react with Magnesium (Mg)?
Which of the following has the greatest ionic character in its bonds?
The electron configuration of Cd2+ is best described by which of the following?
Photo attributed to ScientificRelevance.
The isoelectric point of glycine is 6.0. When glycine is in a buffer with a pH of 6.0, which form predominates?
What is the major product of the reaction below, and what is the mechanism by which it is produced?
(CH3CH2)3CBr + F– –> ; temperature: 500C; solvent: water
[php snippet=1]
A 13 gram sample of an unknown hydrocarbon is a gas at room temperature (25 °C) and atmospheric pressure. Its volume is 11.2 liters. Which of the following could the hydrocarbon be? Note: the ideal gas constant is 0.082 (L*atm)/(K*mol)
[php snippet=1]
Which of the following compounds has the highest boiling point?
[php snippet=1]
Which of the following correctly expresses both a correct statement and a correct explanation for that statement?