Highest Heat of Combustion
[php snippet=1]
Which of the following molecules have the highest heat of combustion per CH2?
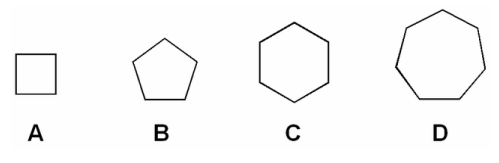
[php snippet=1]
Which of the following molecules have the highest heat of combustion per CH2?

[php snippet=1]
The technique of fractional distillation is most effective for:
[php snippet=1]
A radioactive substance A was placed in a lead-lined container and the radiation it emitted was allowed to pass through a small aperture in the container and below a positively charged plate. The path of the particle changed such that it angled up toward the plate. Which of the following could it be?
[php snippet=1]
The following reaction was carried out in a closed system:
N2(g) + O2(g) <--> 2NO(g)
 Hrxn = -ve value
Hrxn = -ve value
Which of the following will favor the production of NO?
[php snippet=1]
An amino acid in a medium with a pH value above its isoelectric point would have:
[php snippet=1]
Which of the following is NOT characteristic of hydrogen bonding?
[php snippet=1]
238U is radioactive. One of the intermediates in its decay is obtained via 3 alpha emissions, 2 beta emissions and 3 gamma emissions. What is the identity of this intermediate? (You may consult a Periodic Table)
[php snippet=1]
At a given temperature T in kelvin, the relationship between the three thermodynamic quantities including the change in Gibbs free energy (![]() G), the change in enthalpy (
G), the change in enthalpy (![]() H) and the change in entropy (
H) and the change in entropy (![]() S), can be expressed as follows:
S), can be expressed as follows:
The sublimation of carbon dioxide occurs quickly at room temperature. What might be predicted for the three thermodynamic quantities for the reverse reaction?
[php snippet=1]
Each of the following structures is a resonance form of the molecule shown below EXCEPT one. Which one is the EXCEPTION?
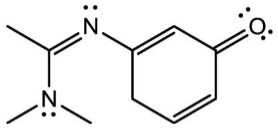
[php snippet=1]
Enzyme X creates peptide bonds between amino acids leucine and valine, as well as peptide bonds between leucine molecules and between valine molecules. If enzyme X catalyzed the production of leu2val2, how many different linear tetrapeptides would be possible?